
Question
The distance between the centres of equal circles each of radius 3cm is 10cm.the. Length of a transverse tangent is?
The sum of first three terms of a G.P. is 12
13
and their product is –1. Find G.P
Conduct a survey at population in your locality taking a sample of atleast 50 families. Prepare
a project report with respect to the following points:
(i) Represent the data showing age using class intervals.
(ii) Compute the average income of the families.
(iii) Literacy level of the population in terms of Elementary, Secondary, Senior Secondary,
Graduation and above.
(iv) Find the variance of the income of the families.
(v) Represent the data in part (iii) with the help of bar graph.
draw two concentric circles with centre o and radius 3 cm and 3.6 cm.
Can anyone tell me the tds rate for propritorship professional charges?


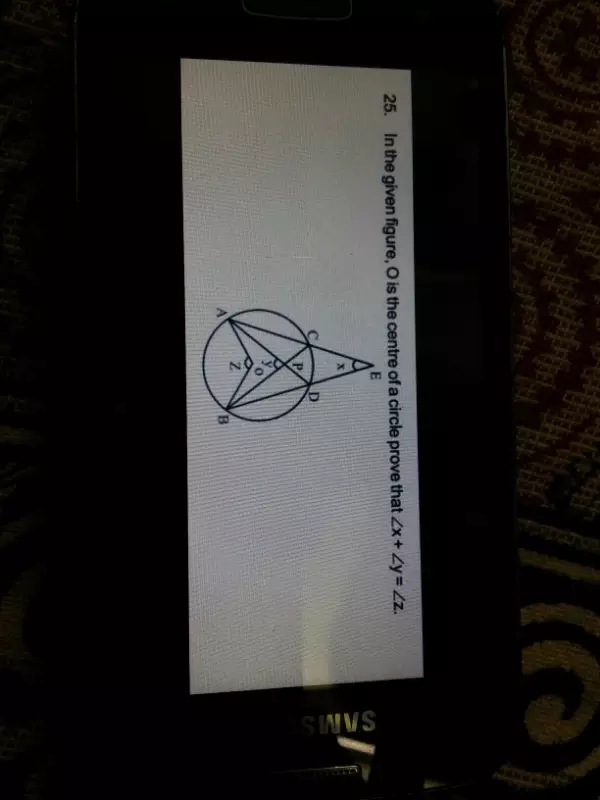 prove that x y=z
prove that x y=z